Benefit from a robust mixed leukocyte reaction assay to evaluate immunomodulatory properties of your candidates
12 / 04 / 2023

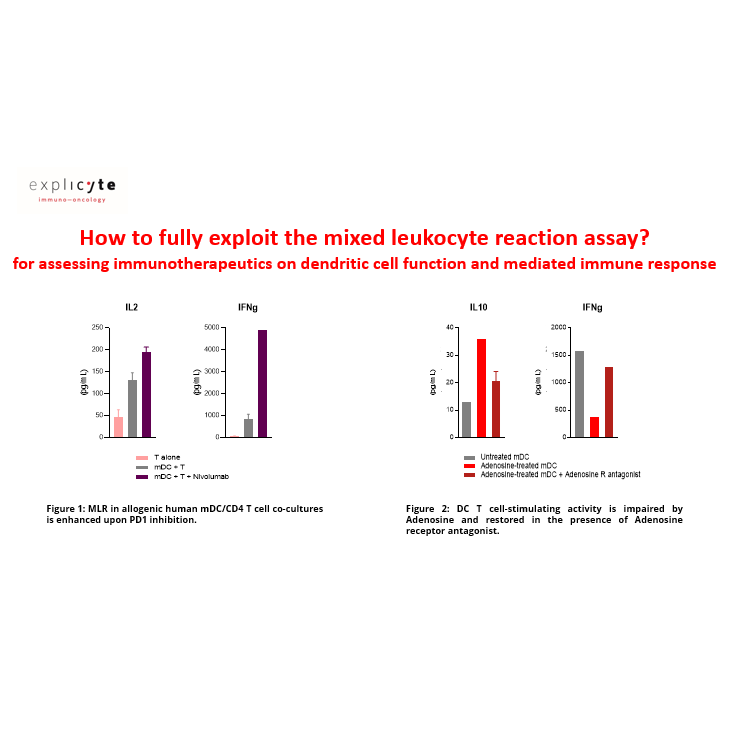
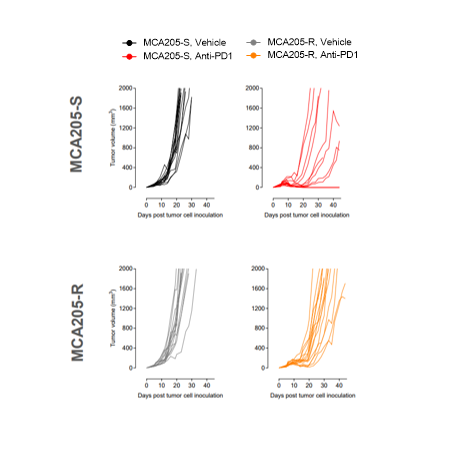
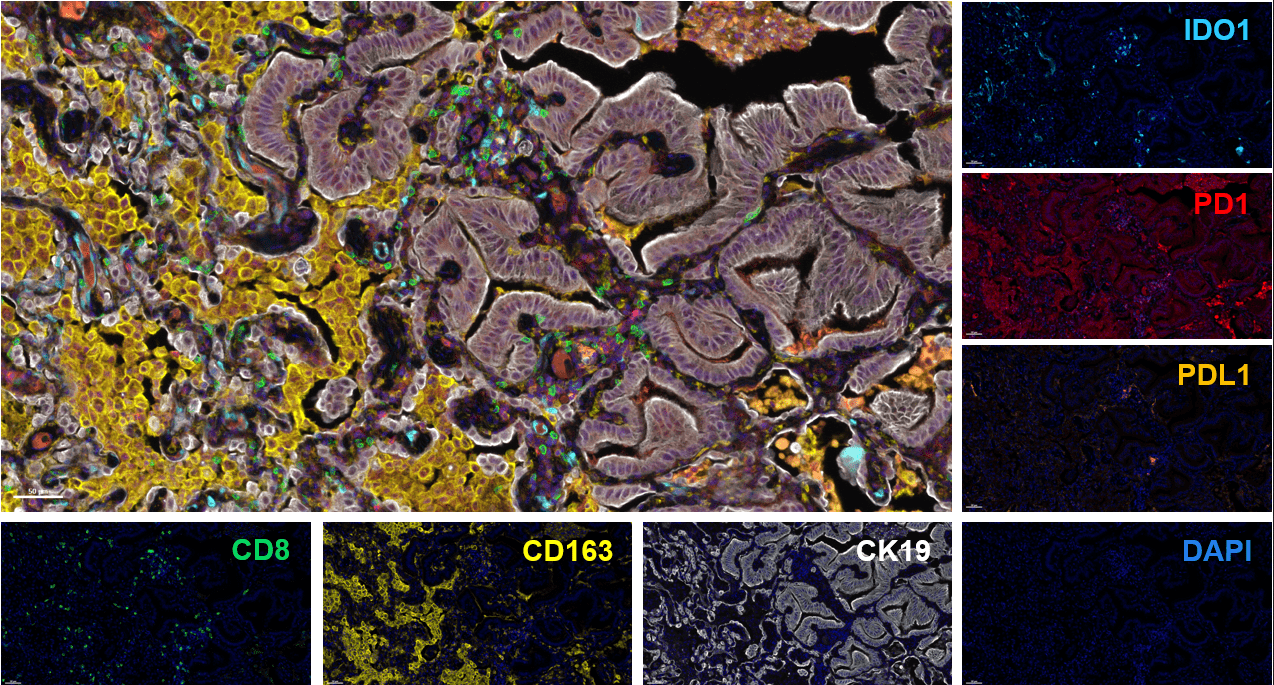
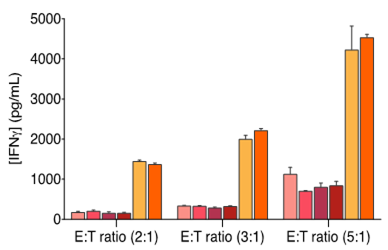
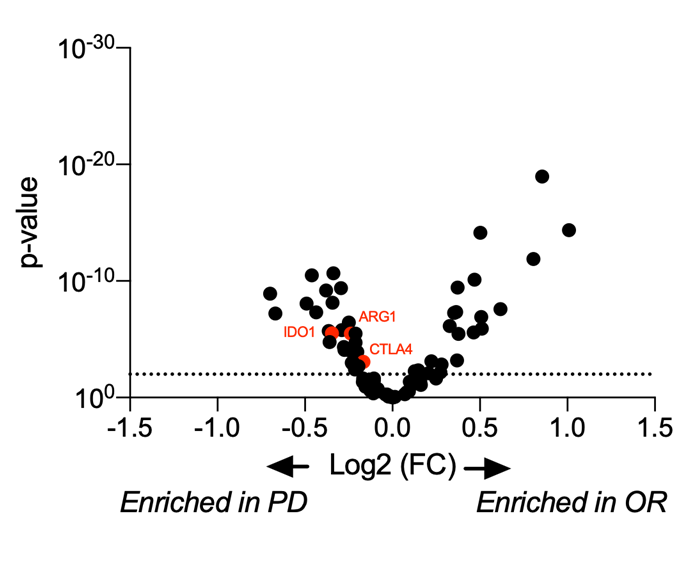
Considering the importance of dendritic cells in fine-tuning T cell response, their manipulation represents an attractive approach to elicit or boost an anti-tumor immune response. To this end, we have developed and validated a mixed leukocyte reaction (MLR) assay to address DC function and evaluate their immune-modulatory properties.