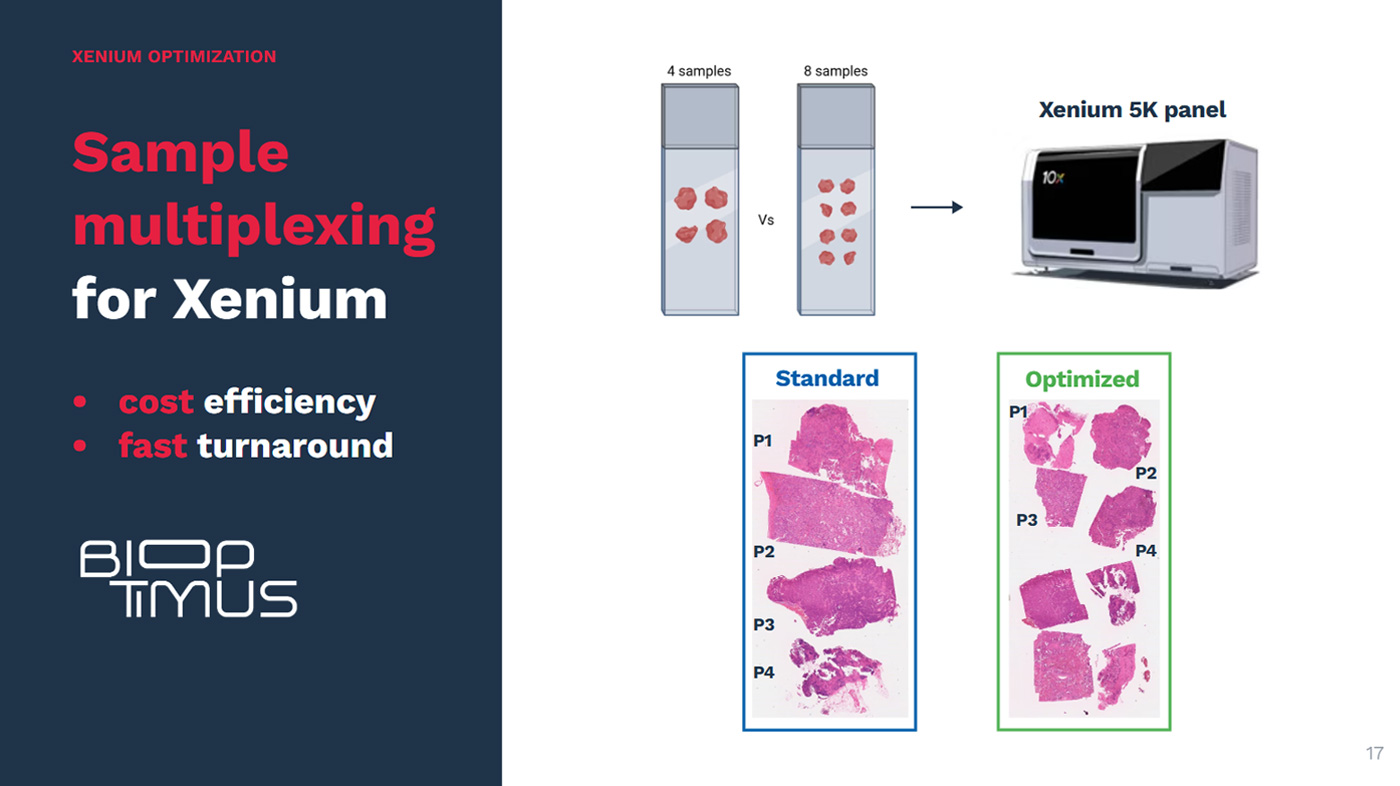
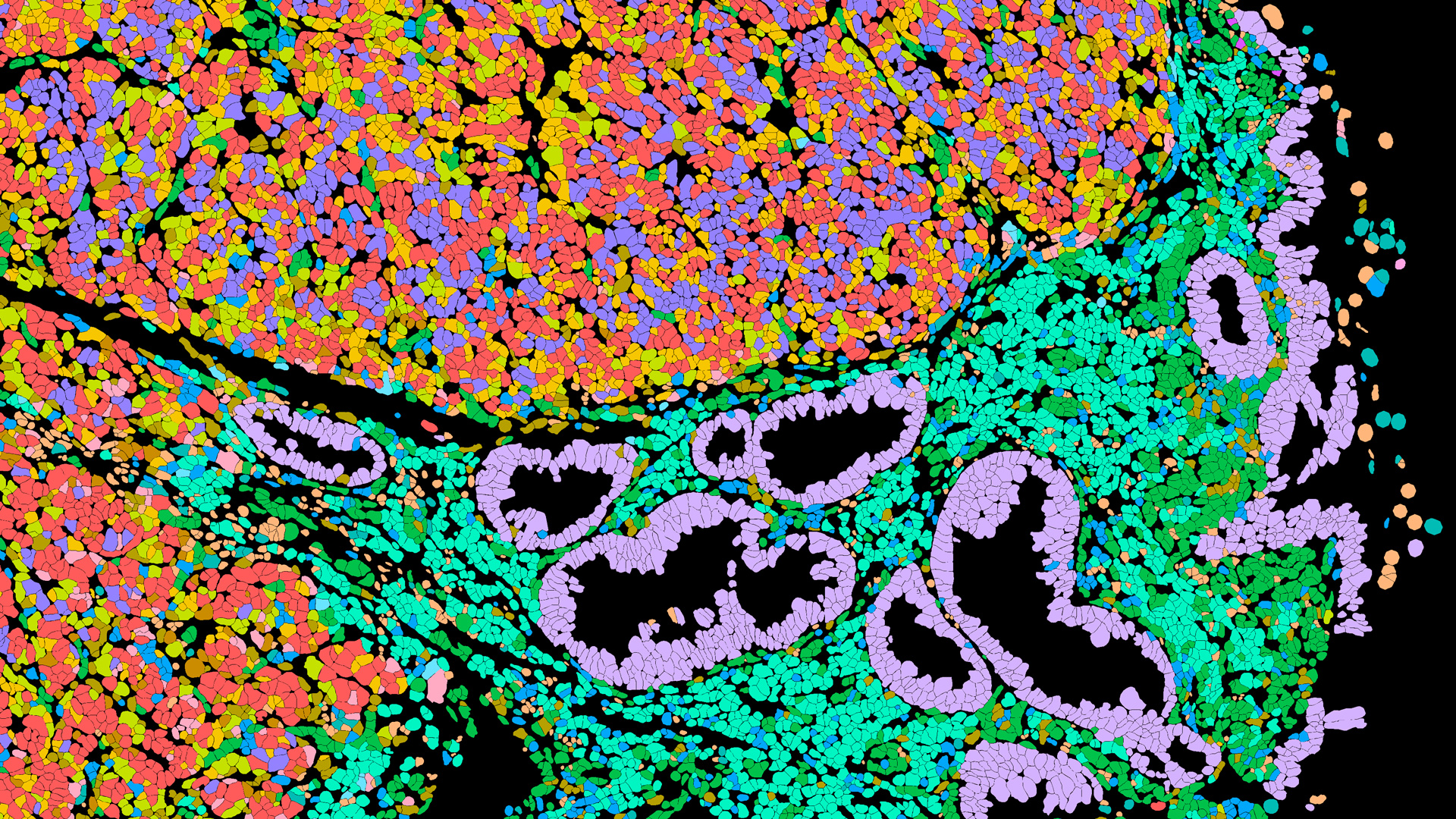
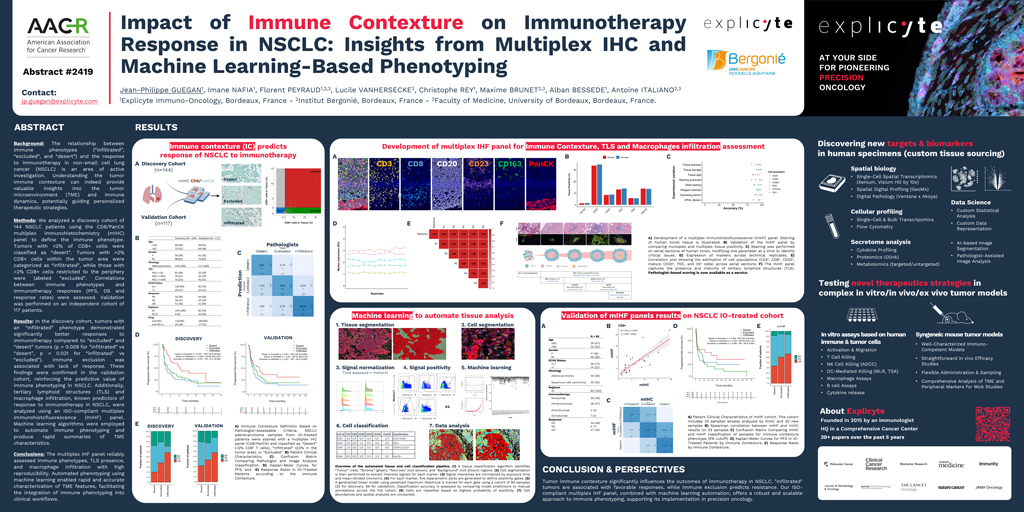
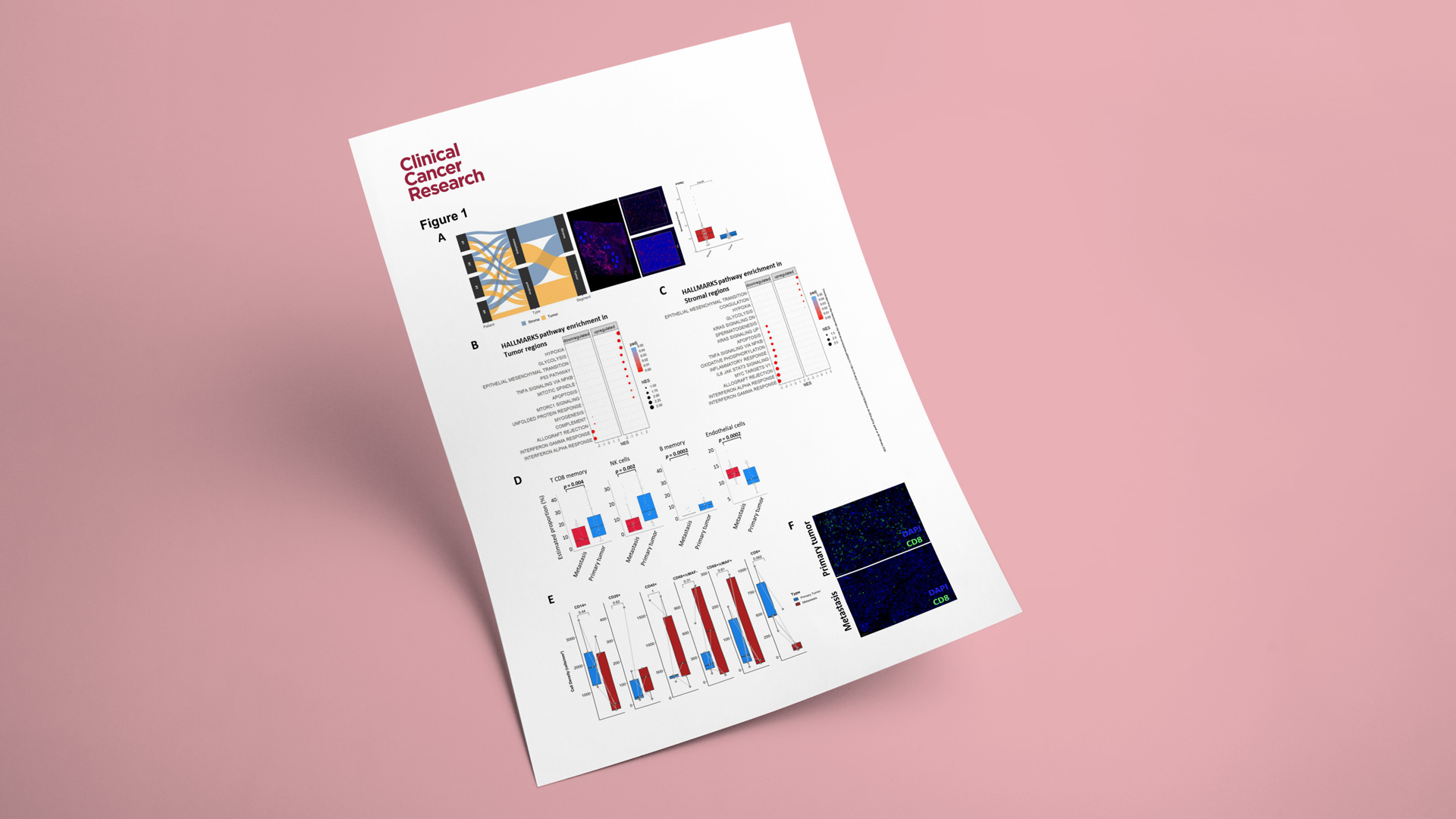
Predicting which NSCLC patients will benefit from immunotherapy requires more than measuring a single biomarker. Our previous work has helped show that key spatial features of the tumor microenvironment—such as
mature tertiary lymphoid structures (
Nature Cancer, 2021;
Cell Reports Medicine, 2025) and
macrophage infiltration within the tumor compartment (
Journal for ImmunoTherapy of Cancer, 2022)—are closely associated with treatment outcome.
Building on these findings, we developed and validated a multiplex immunofluorescence (mIF) panel that integrates
immune contexture, TLS presence and maturity, and macrophage infiltration in a single assay. First presented at the
2025 AACR Annual Meeting, this panel is designed to support translational research and early-phase trials in NSCLC.
Why immune contexture matters
Immune checkpoint inhibitors have transformed cancer treatment, but responses remain highly variable. One reason is that the anti-tumor immune response depends not only on whether immune cells are present, but also on
where they are located. This is the principle behind immune contexture.
Tumors can be broadly grouped into three phenotypes based on CD8 spatial distribution:
- Infiltrated tumors contain CD8+ T cells within the tumor area.
- Excluded tumors show CD8+ T cells at the periphery, without effective penetration into the tumor parenchyma.
- Desert tumors display little to no CD8+ T-cell presence.
This spatial organization is informative because it reflects distinct mechanisms of anti-tumor immunity, immune suppression, or immune escape. Immune contexture does more than label tumors as “hot” or “cold”: it helps explain whether T cells are present, whether they can reach cancer cells, and whether the local microenvironment is permissive or suppressive.
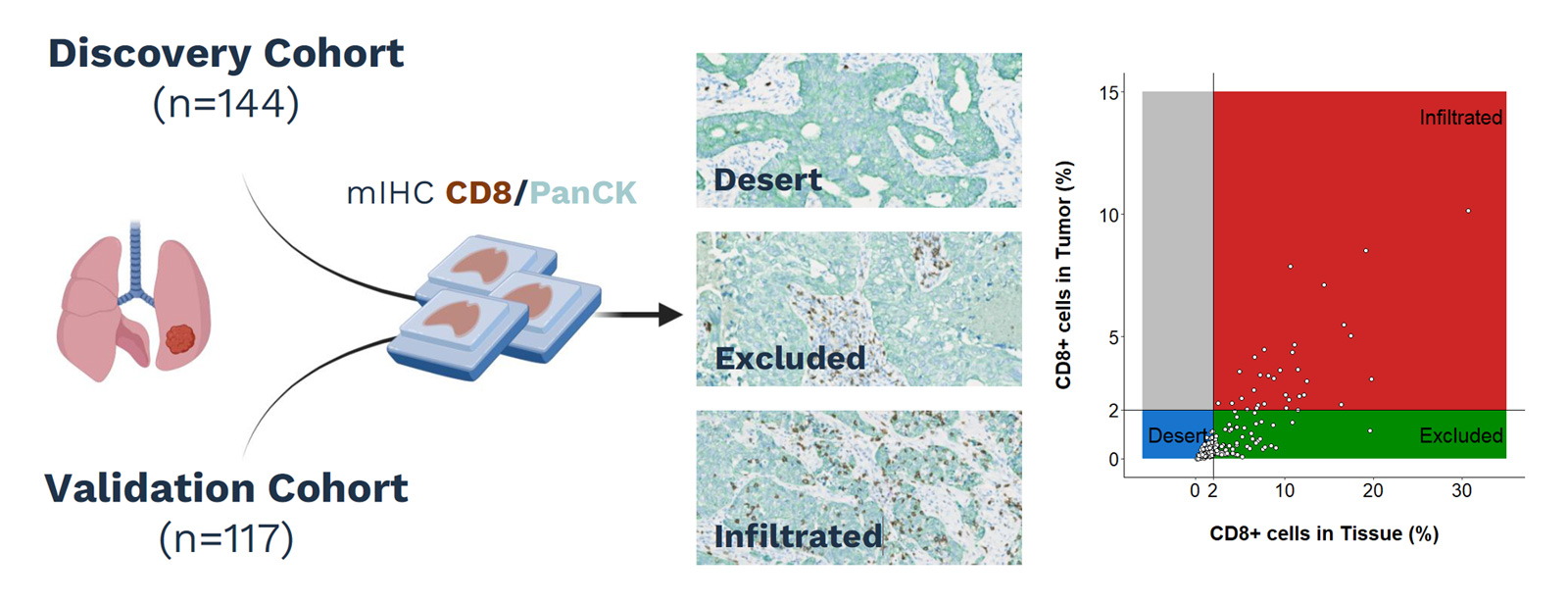
Using a
CD8/PanCK multiplex IHC panel, our AACR poster showed that infiltrated, excluded, and desert phenotypes are associated with different outcomes under immune checkpoint blockade in NSCLC. In both the discovery and validation cohorts, infiltrated tumors were associated with better responses, while immune exclusion was linked to resistance. These results reinforce the value of immune contexture as a spatial biomarker for patient selection.
Why combine it with TLS and M2 macrophages
Immune contexture is highly informative, but it does not capture the full complexity of the tumor microenvironment. To obtain a broader and more actionable spatial readout, we combined it with two additional components that are strongly linked to immunotherapy response in NSCLC: tertiary lymphoid structures (TLS) and M2-like tumor-associated macrophages.
Tertiary lymphoid structures (TLS) reflect the capacity of the tumor microenvironment to support local immune activation. By enabling antigen presentation, lymphocyte priming, and coordinated immune responses, they can favor effective anti-tumor immunity and improve sensitivity to immune checkpoint inhibitors. Their presence, proximity to the tumor, and especially their maturity, therefore provides important information on the quality of the local immune response. This is consistent with the
Nature Cancer publication we co-authored in 2021, which identified
mature TLS as a predictive feature of immunotherapy efficacy.
M2-like tumor-associated macrophages are major drivers of immunosuppressive signaling within the tumor microenvironment. By promoting tissue remodeling, dampening cytotoxic immune responses, and contributing to barriers against effective T-cell activity, they can support resistance to immunotherapy. Their spatial distribution within the tumor compartment therefore helps capture a distinct mechanism of immune escape that complements immune contexture analysis. This is supported by our 2022
Journal for ImmunoTherapy of Cancer publication on
macrophage infiltration and immunotherapy response in NSCLC.
By integrating these three biomarkers into a single panel, we can generate a more complete spatial readout of the tumor immune landscape than with any marker alone.
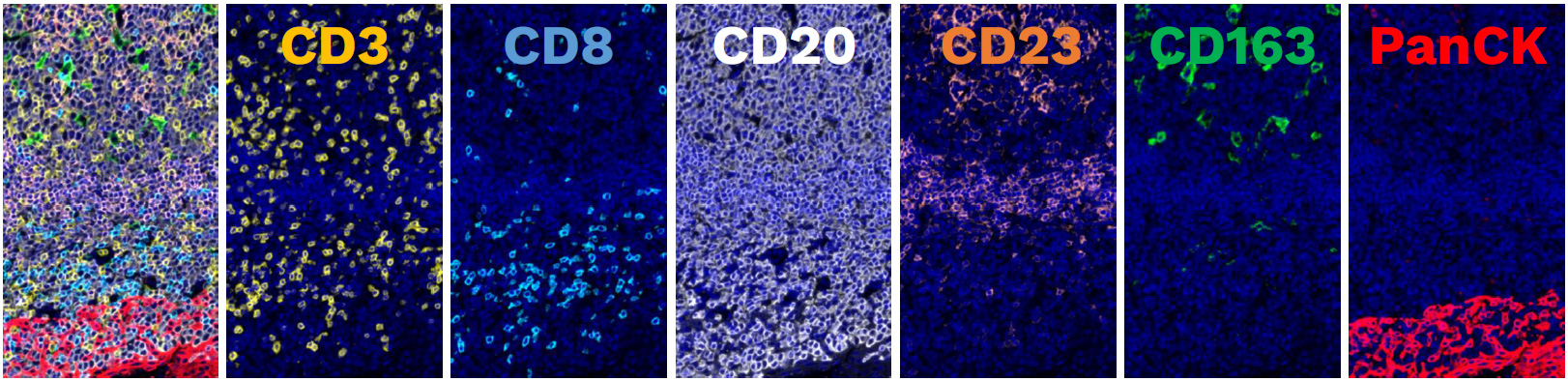
A panel designed for spatial biology at scale
One of the strengths of this approach is that it goes beyond staining alone. The panel is paired with an automated tissue analysis workflow that includes tissue segmentation, cell segmentation, signal normalization, marker positivity scoring, machine learning-based classification, and downstream spatial analysis. This enables complex spatial information to be translated into standardized outputs with pathologist-guided interpretability and high reproducibility.
Our AACR poster also showed concordance between this broader mIF assay and immune-contexture classification derived from the CD8/PanCK multiplex IHC approach in an NSCLC immunotherapy-treated cohort (N=60). Together, these results support the robustness of the panel for translational use and its compatibility with scalable tissue analysis workflows.
Download our 2025 AACR poster
Applications for translational research and early-phase trials, beyond NSCLC
Implemented within Explicyte’s
ISO 13485- and ISO 9001-certified quality framework, this integrated panel is particularly relevant for translational research and early-phase trials in precision oncology. It can support
biomarker discovery, patient stratification, responder versus non-responder analyses, mechanism-of-action studies, and exploratory enrichment strategies in immunotherapy trials.
Although developed in NSCLC, the same approach may be valuable in other indications where spatial immune organization is likely to influence response, including head and neck cancer, urothelial carcinoma, melanoma, renal cell carcinoma, and selected breast or gastrointestinal tumors. Sarcoma is also a relevant setting, as illustrated by the
PEMBROSARC TLS-positive soft-tissue sarcoma study published in Nature Medicine in collaboration with Institut Bergonié and Gustave Roussy.
Interested in bringing spatial immune profiling into your precision oncology program? Contact our team to discuss how this panel could support your next study.
https://youtu.be/bmokMO6jCHc?si=6tz9CLxEy1R_0uHo