Target identification & validation using preclinical & clinical samples
- Tissues & liquid samples provided by the sponsor or sourced by Explicyte
- Fit-for-purpose technology platforms, including digital pathology, single-cell & spatial transcriptomics, FACS, proteomics …
- Expertise in data science to make the most of your dataset

Preclinical profiling for mechanism of action studies
- Samples obtained from external in vivo platforms or provided by the sponsor
- Validated approaches to detect tumor microenvironment (TME) markers & development of custom panels to highlight the target of interest
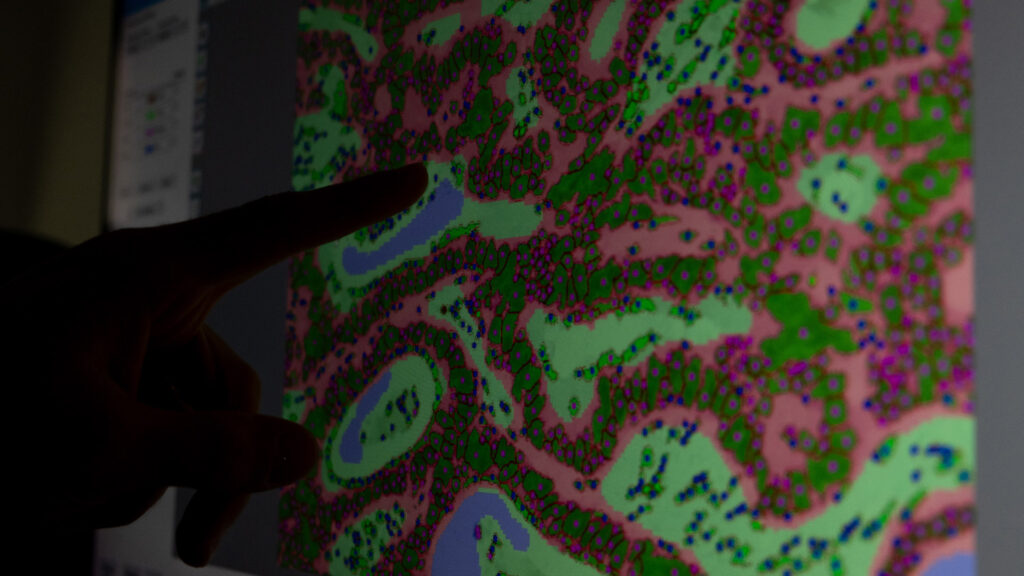
Biomarker research & biomarker testing in clinical samples
- Large capacities to process and analyze patient samples from clinical trials & retrospective studies
- Solid track record in biomarkers studies >20,000 patient samples already analyzed
Why working with Explicyte?
Experts
in Immuno-Oncology
- A comprehensive platform to generate robust datasets
- Over 10 years of experience in TME & peripheral marker profiling
- 30+ peer-reviewed publications in high-impact immuno-oncology journals
Personalized
approach
- Scientific interactions to discuss your project needs (e.g. nature of samples, biomarker panels, etc.)
- A dedicated study director (PhD) from experimental plan to final report discussion
- In-house data science team for rapid dataset analysis
Your contacts

Talk to our team !
Paul Marteau, PharmD (preclinical study director), Imane Nafia, PhD (CSO), Loïc Cerf, MSc (COO), Alban Bessede, PhD (founder, CEO), Jean-Philippe Guégan, PhD (CTO)