We will be attending the AACR Virtual Annual Meeting, from June 22 to 24, 2020 to present our posters featuring our capacities / platforms that support drug discovery in Immuno-Oncology.
Anti-angiogenic therapy combination with hypoxia-regulating agent leads to improved tumor regression in a murine model of renal carcinoma
Presenter/Authors
Abstract
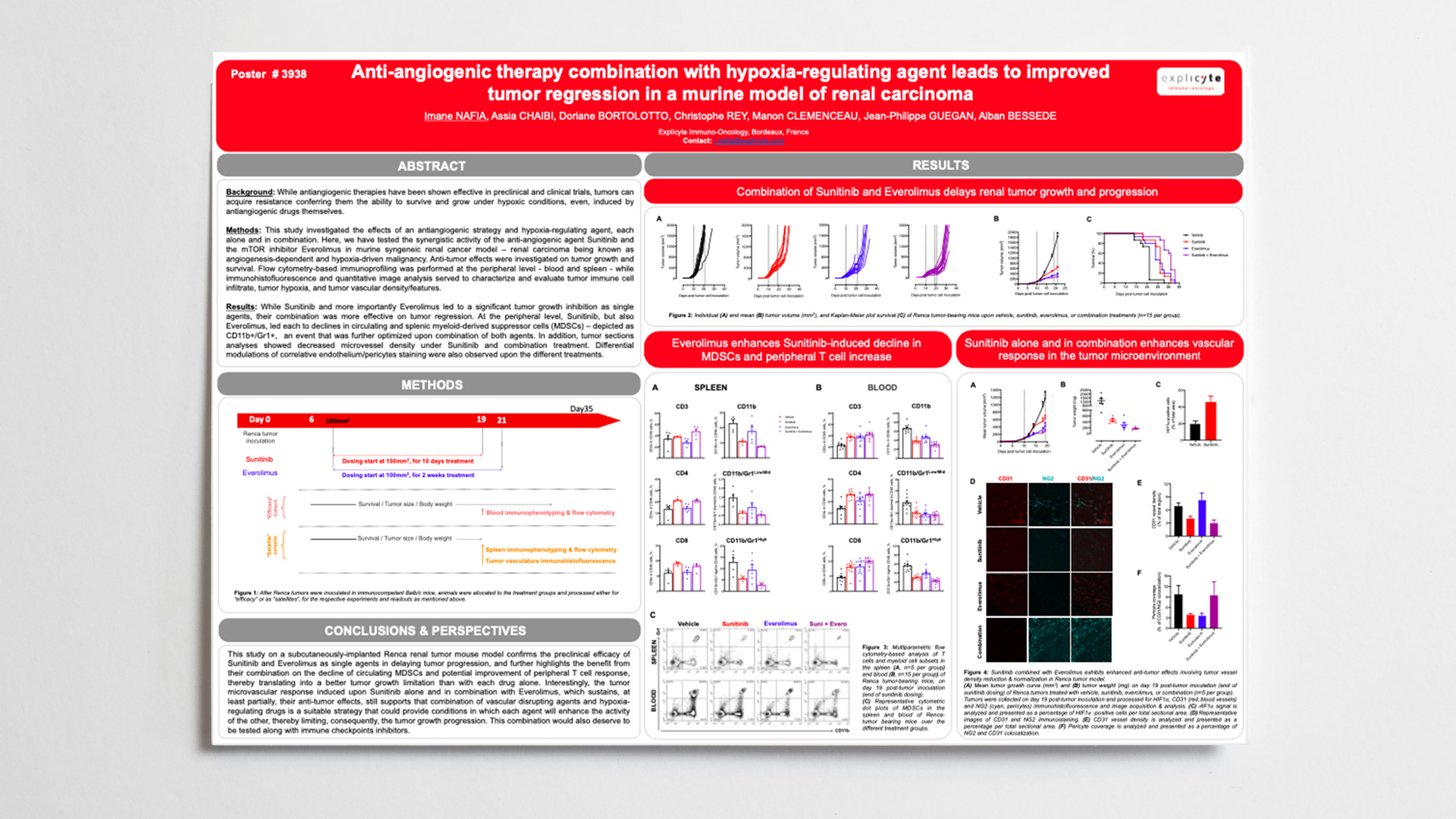
Methods: This study investigated the effects of an antiangiogenic strategy and hypoxia-regulating agent, each alone and in combination. Here, we have tested the synergistic activity of the anti-angiogenic agent Sunitinib and the mTOR inhibitor Everolimus in murine syngeneic renal cancer model – renal carcinoma being known as angiogenesis-dependent and hypoxia-driven malignancy. Anti-tumor effects were investigated on tumor growth and survival. Flow cytometry-based immunoprofiling was performed at the peripheral level – blood and spleen – while immunohistofluorescence and quantitative image analysis served to characterize and evaluate tumor immune cell infiltrate, tumor hypoxia, and tumor vascular density/features.
Results: While Sunitinib and more importantly Everolimus led to a significant tumor growth inhibition as single agents, their combination was more effective on tumor regression. At the peripheral level, Sunitinib, but also Everolimus, led each to declines in circulating and splenic myeloid-derived suppressor cells (MDSCs) – depicted as CD11b+/Gr1+, an event that was further optimized upon combination of both agents. In addition, tumor sections analyses showed decreased microvessel density under Sunitinib and combination treatment. Differential modulations of correlative endothelium/pericytes staining and hypoxia degree were also observed upon the different treatments. This study confirms the preclinical efficacy of Sunitinib and a hypoxia regulator in renal tumor model and suggests that limitation of Sunitinib-induced hypoxia through mTOR targeting or with hypoxia regulators represents a suitable strategy to enhance effect of anti-angiogenic agent thereby limiting tumor growth
Learn more our Renca model