How about an opportunity to assess your compounds in a comprehensive and cost-effective manner?
As case study presented here, combination of multiparametric platforms e.g. intratumoral microdialysis and flow cytometry allows to highlight intrinsic features of the model itself, and to show that PDL1 blockade promotes a strong antitumor activity, an effect driven at least through the relieve of specific immunosuppressive pathways and promotion of effector CD8 T cells.
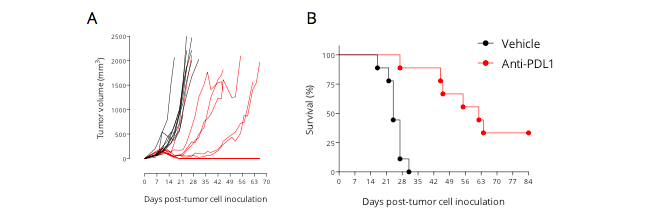
Figure 1: Subcutaneously-implanted MCA205 sarcoma tumor-bearing mouse model is responsive to PDL1 blockade. Mice were subcutaneously implanted with tumor cells and exposed to anti-PDL1 antibody. Tumor growth was monitored overtime via calipering. Tumor volume (A) and survival (B) were then determined.
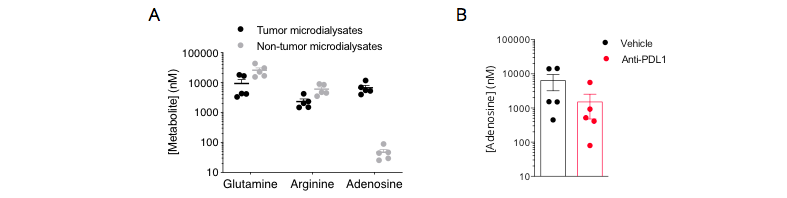
Figure 2: Immunometabolic profiling of the MCA205 sarcoma model by intratumoral microdialysis in control and PDL1 blockade conditions. Mice were implanted with tumor cells and then processed for intratumoral microdialysis at the tumor volume average of ~200mm3. (A) Determination of glutamine, arginine, and adenosine levels by LC/MS. The tumor model displays differential levels for arginine and particularly for adenosine between tumor and non-tumor compartments – both pathways are known to be involved in the immune escape. While being essential for a proper immune system functioning, glutamine and arginine levels were shown to be lower, while adenosine was shown to be higher, within the tumor microenvironment, thereby conferring a peculiar immunometabolic profile to this model. (B) PDL1 blockade triggers a decrease of tumoral adenosine production. Adenosine levels in tumor microdialysates of MCA205-bearing mice treated or not with anti-PDL1 were measured by LC/MS. Anti-PDL1 induced a limitation of the tumoral adenosine level thus arguing for an important role of the adenosine axis in the control of the anti-tumor immune response.
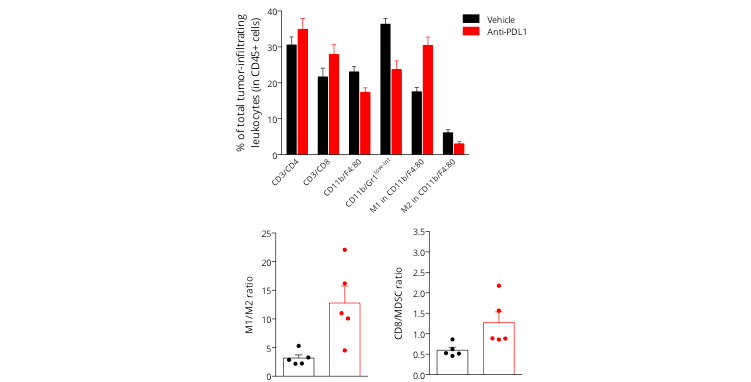
Figure 3: Analysis of PDL1 blockade mechanism of action on the MCA205 tumor-infiltrating leukocytes populations. Flow cytometry analysis of MCA205 tumors shows infiltration by both lymphocytic (CD4, CD8) and myeloid cells including the immunosuppressive MDSC population (CD11b/Gr1low-int) and macrophages (M1 and M2 subsets). Upon anti-PDL1 treatment, investigation of tumor-infiltrating leukocytic cell subsets showed an increase in CD8 cytotoxic lymphocyte proportion as well as a decrease of MDSC cell infiltration. In addition, while vehicle tumors were shown to be infiltrated by M2 macrophages, which helps conferring an immunosuppressive microenvironment, PDL1 blockade was demonstrated to reprogram this macrophage profile by increasing the M1 and decreasing the M2 subset proportions, thereby suggesting an effect on macrophage polarization and proliferation.
Shuttle for efficacy assessment on 6-week experimental session
- Model / Strain: According to the Sponsor’s requirements
- Readouts: Tumor growth / Body weight / Survival
- Standard reference: Immune checkpoint inhibitor – to be discussed with the Sponsor
- Group size: At least 10 mice per group
Optional analyses:Satellite studies for immune response profiling and MoA delineation (Intratumoral microdialysis/metabolite quantification, flow cytometry, gene expression analysis, immunohistochemistry…).
More information about our in vivo services