Glioblastoma (GBM) is the most aggressive primary brain tumor. Despite multimodal treatment strategies with surgery, radiation therapy and chemotherapy, the prognosis of GBM patients remains dismal. GBM are characterized by a high immune cells infiltration including myeloid derived suppressor cells (MDSCs), microglia or regulatory T cells (Tregs), all of them contributing to the creation of an immunosuppressive micro-environment. While manipulating the immune system to restore its anti-tumor activity have shown efficacy in many cancers, immunotherapy figures as an attractive therapeutic option for glioblastoma and several clinical trials are currently ongoing.
To support your R&D program(s) in glioma, Explicyte has validated a syngeneic intracerebral mouse tumor model of Glioblastoma on which gold standard chemotherapy (Temozolomide, TMZ) and immunotherapy (CTLA4) has been validated. We thus propose to evaluate the efficacy of your candidate compound(s) – on its own or combined with a reference treatment – by mean of bioluminescence imaging assessment (on a weekly basis) and close monitoring of survival (on a daily basis).
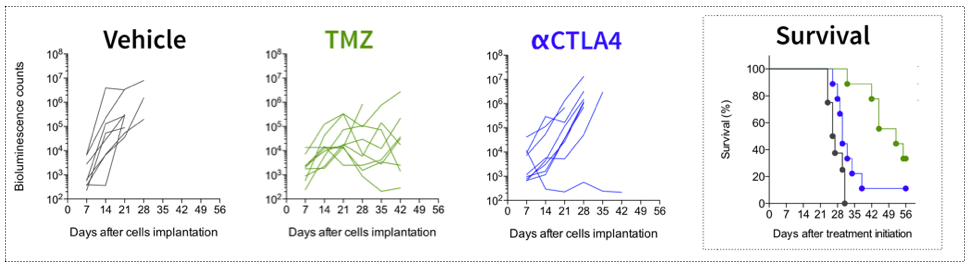
Effect of Temozolomide and Immune checkpoint inhibitors on tumor growth (bioluminescence imaging) and survival of GL261-Luc2 (glioblastoma) tumor bearing mice. Mice were implanted with GL261-Luc2 tumor cells, and exposed either to Temozolomide (TMZ), or anti-CTLA4 antibody. Tumor growth was followed over time and determined by in vivo bioimaging.
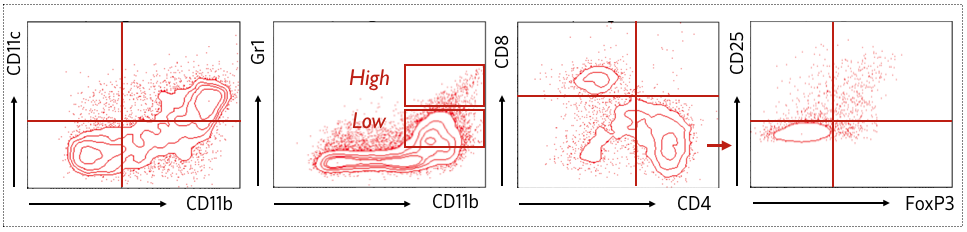
Also, to strengthen the efficacy study, and characterize the underlying mechanism of action, ancillary analysis can be implemented such as isolation and characterization of brain infiltrating lymphocytes (BILs) by high throughput flow cytometry (MDSCs, Treg, Teff, DCs, etc) or immunohistochemistry analysis.
Example of immune profiling using FACS. Characterization of BILs by flow cytometry revealed the presence of myeloid cell subsets – distinguished with CD11c+, CD11b+, Gr1High or Gr1Low stainings – and the presence ofregulatory T cells (FoxP3+ / CD25+) within the CD4+ T cells subpopulation.